MedTech Regulatory Services
Guiding Medical Devices from Innovation to Approval
At Archimedic, Regulatory Strategy is never an afterthought — it’s built into every stage of development.
Navigating FDA pathways takes more than paperwork — it takes foresight, precision, and experience. Our team helps innovators define the right regulatory pathway, prepare for key submissions, and build the evidence needed for approval.
From early Q-Subs to final 510(k) or PMA submissions, we ensure your device is not just developed — it’s designed to earn FDA confidence and reach patients faster.
Our Core Regulatory Capabilities
Regulatory success is more than submitting documents.
At Archimedic, we combine deep regulatory expertise, engineering insight, and go-to-market understanding to create regulatory pathways that reduce risk, accelerate approval, and align with your overall design and commercial strategy. Our team works alongside you from concept through submission.
Our capabilities span the full regulatory spectrum, ensuring that your medical device isn’t just designed well, but is positioned to succeed in review, in production, and in the marketplace.
Regulatory Strategy
Whether you’re developing a medical device or combination product, we help you determine the right pathway (510(k), PMA, De Novo, IDE) and build a strategy to get there. Early strategy alignment reduces surprises and sets your project up for success.
Pre-Submission Support
Early engagement with the FDA can de-risk your entire program. We prepare documentation, organize Q-Sub meetings, facilitate communications, and ensure all discussions are structured to provide regulatory clarity when it matters most.
Standards & Guidance Review
We identify and interpret the critical consensus standards, testing requirements, and guidance documents that apply to your device — then integrate them into the design and development process. This ensures your device meets current regulatory expectations from the ground up.
Regulatory Submission Support
From Q-Submissions (Q-Subs), De Novo requests, PMAs, and 510(k)s, our team prepares, compiles, and QCs submission packages that stand up to FDA review. We also provide guidance on market claims, comparative evidence, and labeling requirements to support regulatory approval and subsequent adoption.
Why Regulatory Strategy Matters
Brilliant innovations stall every year because medical device teams underestimate the regulatory path. FDA expectations, predicate selection, trial design, and evolving guidance can derail even the most promising device.
At Archimedic, we don’t see Regulatory as a final checkpoint — we see it as the roadmap that guides development from the start. By embedding regulatory strategy into design and development, we help innovators minimize surprises, reduce costs, and accelerate time-to-market.
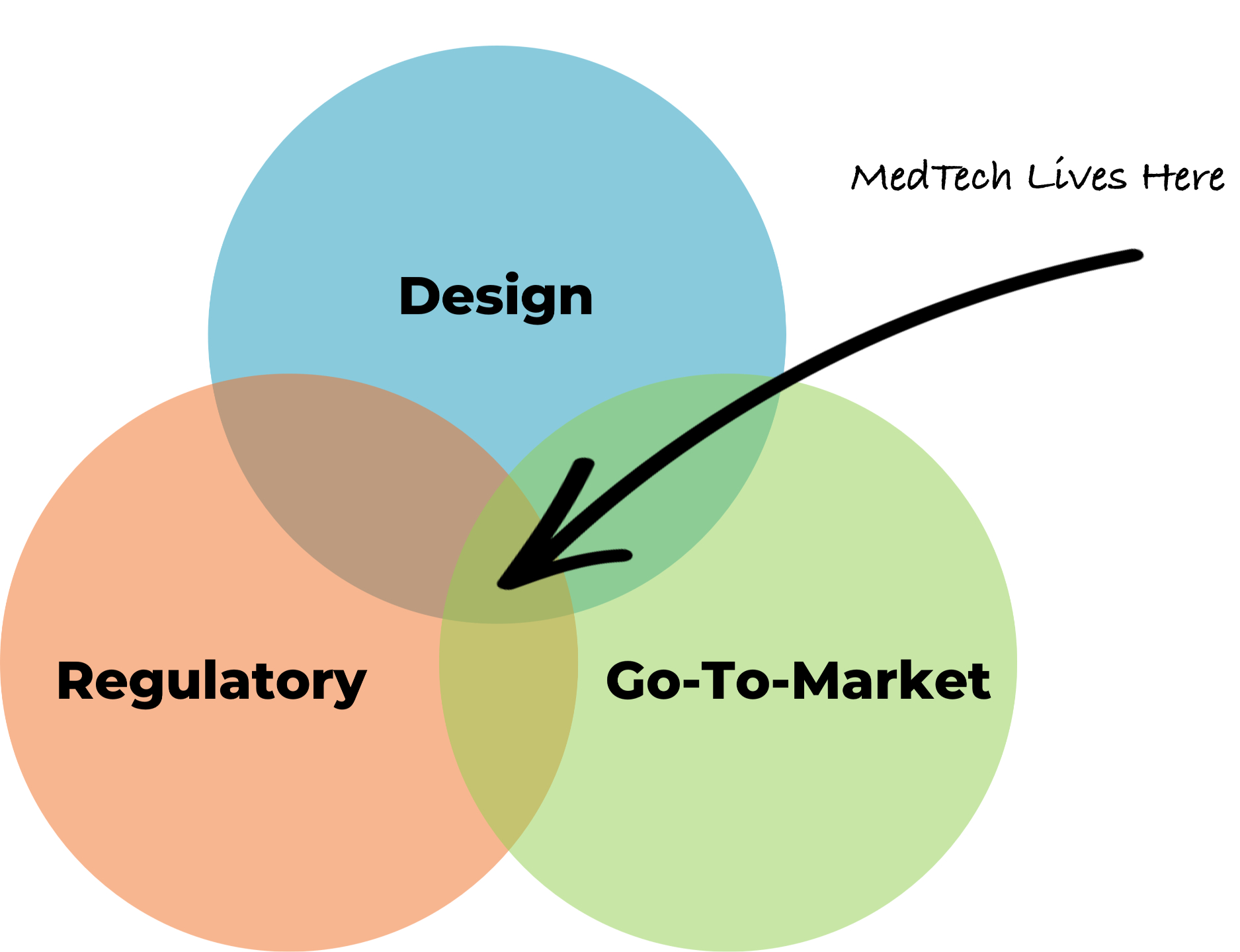
Our integrated Design + Regulatory + Go-to-Market (DRG) approach ensures your device is not only innovative but also compliant, review-ready, and positioned for success in the marketplace
Why Choose Archimedic?
We’re more than your typical medical device firm.
We’re a full-path MedTech partner who understands that medical device success requires integration across Design, Regulatory, and Go-to-market workflows.
- Decades of experience navigating FDA and regulatory pathways for medical devices and combination products.
- Integrated DRG approach that embeds regulatory thinking into design and development.
- Proven track record of successful submissions across device classes and therapeutic areas.
- Collaborative team of engineers, strategists, and regulatory experts working under one roof.
- Flexible engagement models tailored to startups, scale-ups, and established MedTech leaders.
Let’s De-Risk Your Regulatory Pathway
The sooner you engage regulatory expertise, the stronger your chance of success.
At Archimedic, MedTech lives here. Let’s create something extraordinary together.
Schedule a 30-minute meeting with a senior member of the Archimedic team to explore possibilities of working together.
Schedule a 30-Minute Consultation