Partner with Archimedic to streamline design, secure regulatory authorization, and drive market adoption.
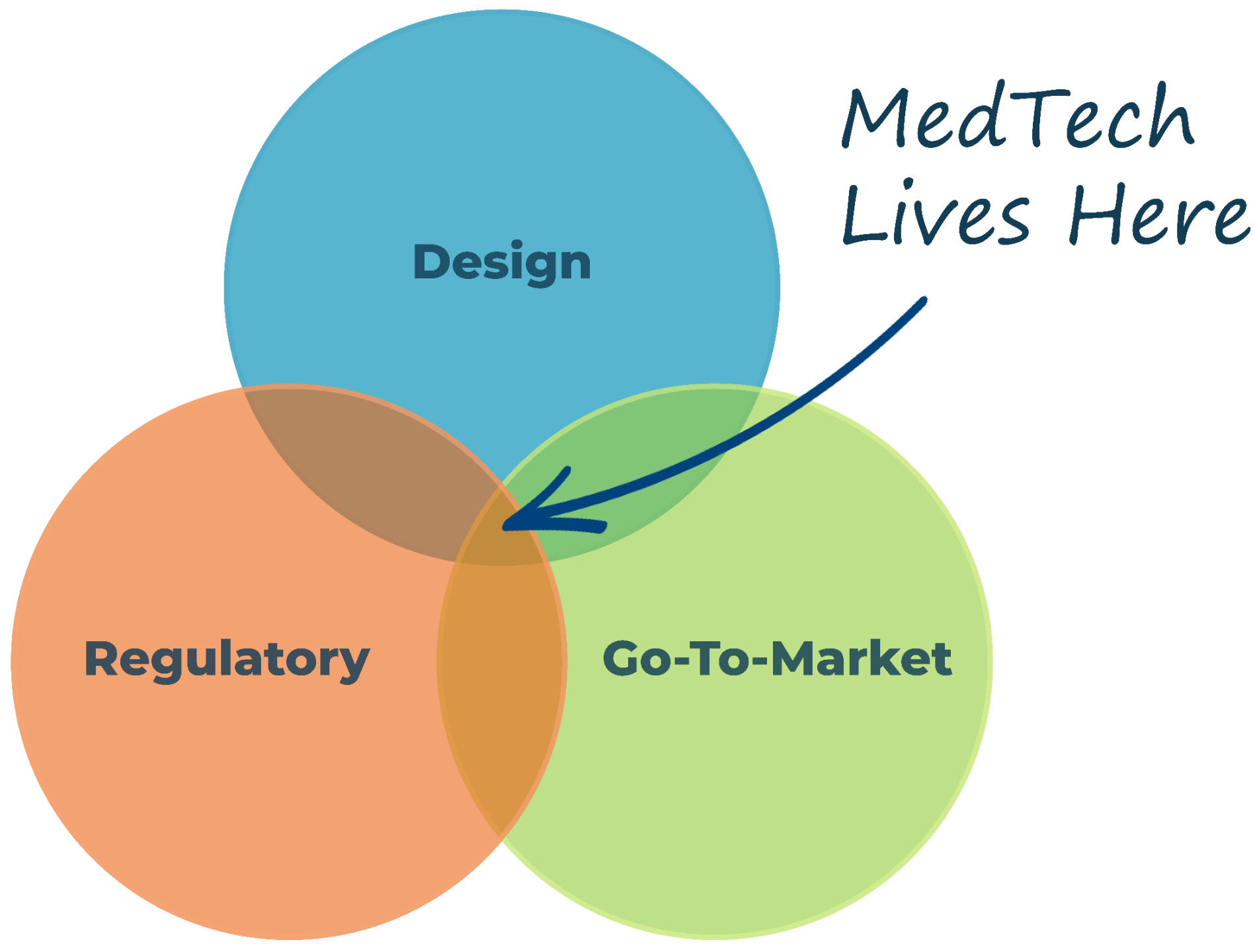
Where Innovation Meets Impact
We go beyond great design to align your device with regulatory pathways and market needs, ensuring it thrives.
How We're Different: Watch the VideoMEDTECH MADE REAL
Case Studies
Learn how Archimedic has helped other MedTech innovators across surgical, drug delivery, diagnostic, and home health categories.
Client Testimonials
What Our Clients Are Saying About Their Experience With Archimedic
Impact Begins with Action
Your mission is clear. Let's connect to shape your device for market success and meaningful change.
Discuss My Project